
Bombarding target stable elements with diverse neutrons accomplished this significant feat. Many of these radioisotopes became produced inside atomic nuclear reactors. The Availability of Radioactive SubstancesĪs knowledge and technology advanced, more and more manufacturers began to supply radioactive materials-naturally and artificially made-for medical applications. Qualified medical researchers and healthcare professionals could obtain batches of radioisotopes-such as the versatile carbon-14-from laboratories and facilities. The incredible buildup of their own research made the distribution of distinctive radioactive isotopic species possible. The discoveries of these intellectual physicists and chemists in the 20 th century significantly influenced the future of medicine-and peacetime applications of atomic energy. Among these notable names are the following: Wilhelm Roentgen, Henri Becquerel, Pierre and Marie Curie, Frederic Joliot, and Enrico Fermi. The fascinating background of radioactivity use for medical purposes is full to the brim with world-renowned scientists and Nobel Prize winners. Here’s a look into how these radioisotopes support research, diagnosis, and therapy in health care. Let’s examine closer the historical applications of 14C in this specialty field. But what about its concentrated medical applications? How is carbon-14 used uniquely in the medical field? Due to the fundamental function of its unique characteristics, this isotope is integral to radiopharmaceutical use in nuclear medicine. The half-life of the radioisotope remains unaltered by external environmental factors, causing the isotope to act as an internal clock to establish the age of assorted objects. The wide majority are aware of this radioisotope’s scientific use for carbon dating. For present-day research, the unmistakably-slow decay rate of carbon-14 is invaluable compared to its common radioactive cousins. The most generally-recognizable isotope is carbon-14 due to its radioactivity, with a notable half-life of 5,730 years. Each is found naturally on Earth, but only one is uniquely unstable. This stable sixth element on the periodic table contains three diverse isotopic forms-carbon 13, carbon-13, and carbon-14. 
Matter-of-fact, an entire branch of chemistry exclusively devotes its studies to carbon-containing compound composition and reaction: organic chemistry.īut there’s more to carbon’s universal story. Many of the millions of identifiable carbon compounds in the world are essential to life as we know it. Nuclear Magnetic Resonance SpectroscopyĬarbon is one of the most abundant elements existing on our planet-one’s existence that humanity’s known about since ancient times. 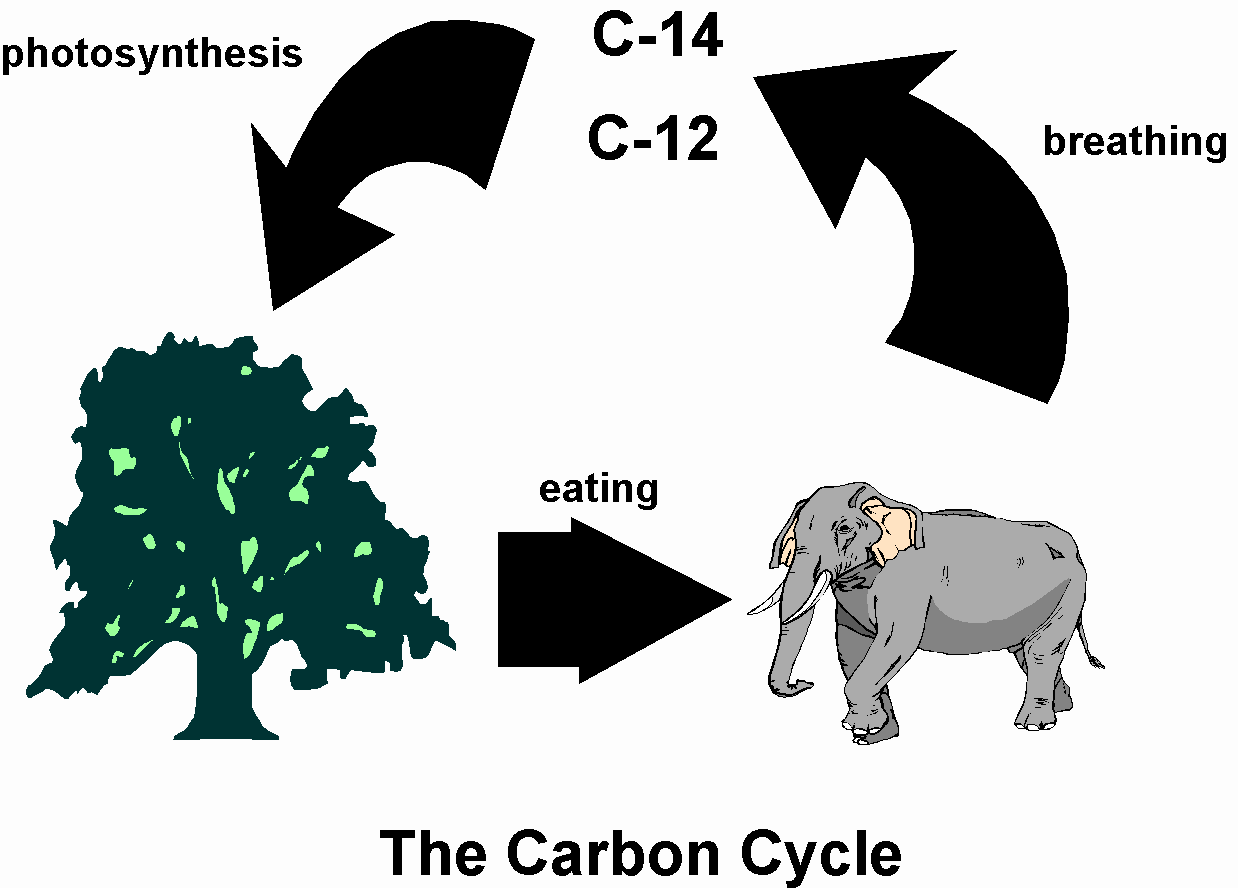
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
